EviGenuity is built on the belief that clarity, structure, and integrity guide every initiative towards a meaningful milestone. We work at the intersection of insight and execution, catering to data driven strategies which move knowledge to actions with purpose and discipline
Our partnership brings evidence closer to actionable insights by bridging the diverse information – creating systems and frameworks that are reliable and scalable.
Summary level outcomes databases
Owing to the ever-increasing challenges in the drug development and product lifecycle, there is a compelling need for rational approaches that contribute to efficient drug development. Model-based meta-analysis is one such critical component that drives informed decision making by leveraging the wealth of published clinical and real-world evidence.
We build comprehensive or customised Summary level outcomes databases that gather published efficacy and safety data from multiple trials across several disease areas structured in a scalable and analysis ready format.
Oncology
Neuroscience
Autoimmune
CV & Metabolic

Pain & inflammation

Vaccines
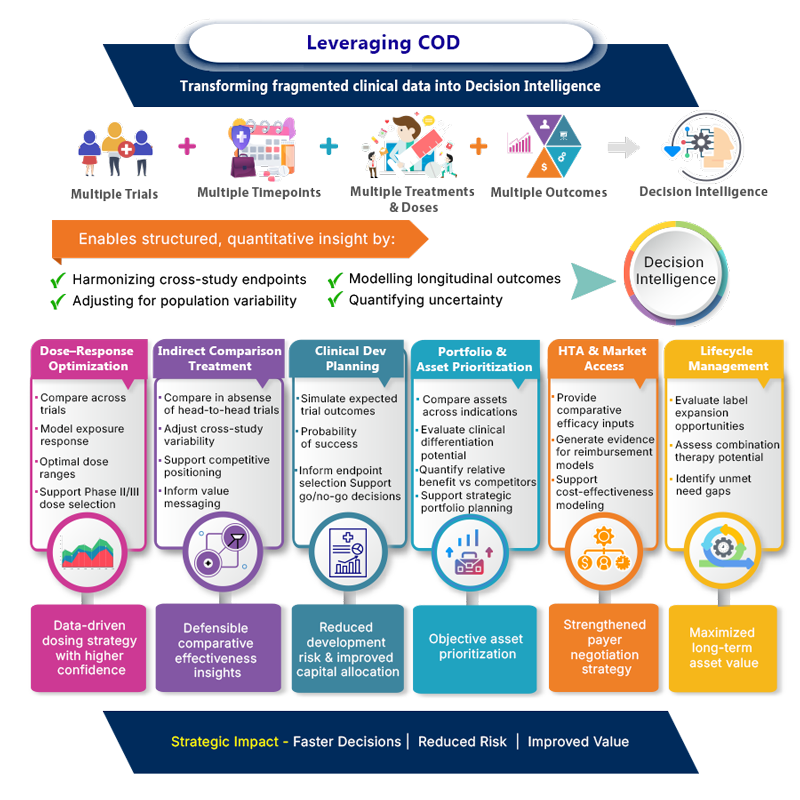
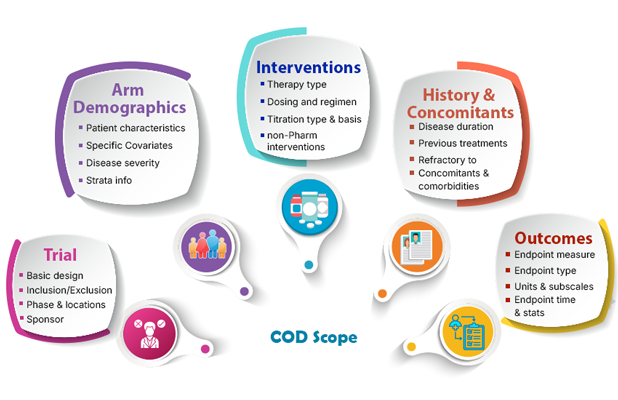
What is COD?
Clinical outcomes databases (COD) are a repository of summary level efficacy and safety endpoints for the entire longitudinal time course data from multiple trails in a given disease area
In order to model the variability in response as an impact of time and treatment, the scope includes the detailed information pertaining to trial design characteristics, patient demographics, strata, baseline covariates, medical histories, concomitant medications and outcome measures with summary statistics and comparison from the trial control.
The length and breadth of the information covered enables normalize the heterogeneity across multiple trials and make meaningful comparisons