Evidence databases are leveraged for high value internal and/or regulatory decisions. It is essential to ensure the data that is plugged for such decision-making is of very high quality and reliability without any bias. The paradigm of the two-tier model of “data provider to Client” does not offer the required validation and confidence on the data.
“EviGenuity” offers an unbiased 3rd party QC with evolved processes, summaries, graphical outputs & quality reports.
- Evidence databases consolidate the information from multiple trails for multiple outcomes. Such published data is highly variable and complex in nature with evolving outcome measures, dosing, scales, subscales, strata/covariates and summary statistics.
- Leveraging extremely variable data for internal and regulatory decision-making demands access to highly reliable data with a well-established processes and unbiased quality benchmarking. Such process involves and entails development beyond multiple stage gates of review
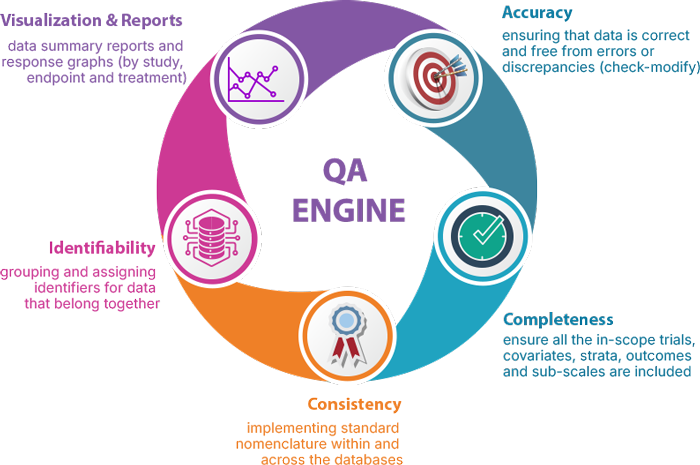
- highly structured and identifiable data
- data standardization with evolving scales and subscales
- normalized and analysis ready data groupings
- Comprehensive quality reports and summaries
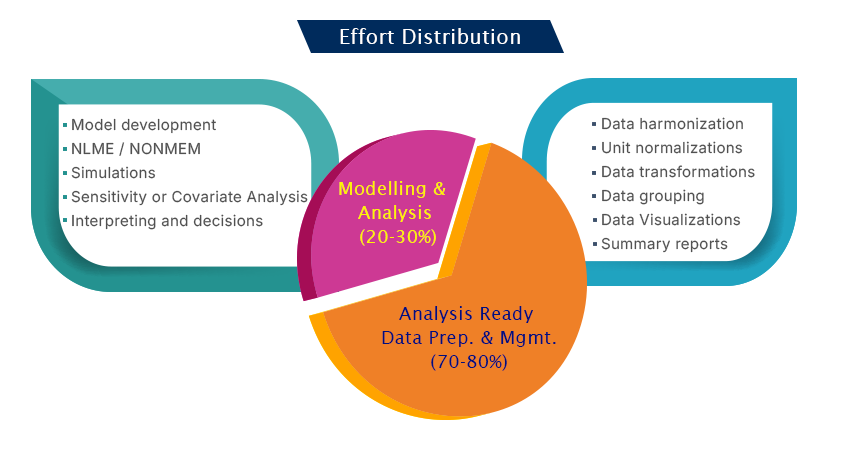
Evidence to decision – Effort distribution
Utilizing the wealth of information for decision making given the variability, is a cumbersome and complex process. Harmonizing the legacy data with the recent trial information is critical and as well time consuming.
As the science continues to evolve, new endpoints and their subscales are frequently encountered as the database undergoes update cycles with new trails. Evigenuity has access to metadata endpoint libraries and right systems to readily implement the standards on such variable data.
Analyst/Pharmacometrician efforts are ideal in transforming the information to knowledge than reviewing and qualifying the data at the stage of analysis.